Document Type : Original Article
Authors
University of North Bengal
Keywords
Introduction
Tea is the most preferred beverage after water and is consumed by almost two-thirds of the world's population (Yen and Chen. 1995, Yang et al. 2022). Tea is an economically important crop in India and North East India accounts for around 75% of overall Tea production in India (Roy et al.2020). There are three regions cultivating tea in North Bengal which are Darjeeling, Terai, and Dooars, and estimated production for November 2022 is 23.47, 19.60, and 0.31 million kg for Dooars, Terai, and Darjeeling, respectively (Tea board of India.2022, https://www.teaboard.gov.in). It empowers the lives of an estimated 11, 00,000 people and more by providing them with livelihood (https://www.thehindu.com/news/national/other-states/tea-industry-gasping-for-survival). Tea production is facing several constraints of which pest attack is the major problem. If pests and diseases aren’t controlled, a 30-50% loss could be observed (Hazarika et al. 2009). More than 1000 arthropods tagged as pests had been recorded in the tea ecosystem (Chen 1989) the world over. The various acarid species that thrive on tea and lead to yield loss are Tetranychus kanzawai Kishida (Acarina: Tetranychidae), Brevipalpus phoenicis Geijskes (Acarina: Tenuipalpidae), Acaphylla theae (Watt) Keifer (Acarina: Eriophyidae), Calacarus carinatus Green (Acarina: Eriophyidae), but, among them, the most common and significant acarid pest is the Red Spider Mite, Oligonychus coffeae Niether (Acarina: Tetranychidae) which was discovered in 1868 in Assam, India (Hazarika et al. 2009). The life cycle of Tea Red Spider Mites is short and they possess a high reproductive capacity ( Das 1959). The Economic Threshold Level of the Tea Red Spider Mite has been determined to be 5 mites per leaf in North East India (Mamun et al.2014). Prolonged application of conventional pesticides and this short-life cycle led to the development of significant pesticide tolerance in the mites (Roy 2019) thereby, leading to a frequent and unmanageable resurgence of the pest which led to crop loss and subsequent economic blow in terms of tea trade in India. Natural products derived from plants are an excellent source of a mixture of active compounds that can effectively control pests in agriculture without developing tolerance to pests (Berenbaum et al. 1991; Feng and Isman. 1995). This favors the use of “Botanicals” that are prepared using the crude extracts of higher plants (Barua 2008). Some of the botanicals that are being used are Pyrethrum, Rotenone, Neem, and essential oils like Menthol from different types of mint species, Thymol from garden thyme (Thymus vulgaris), and Eugenol from clove oil (Syzygium aromaticum) (Isman 2006). India has rich floristic diversity that harbors a wide variety of plants with pesticidal activities. Moreover, Terai and Dooars catalog a wide array of plants with medicinal and toxicological features as reported by Ghosh and Das, 2011. TTRI (Tocklai Tea Research Institute) has started an ITK (Indigenous Technical Knowledge) study as part of IPM, and they have identified 24 native plant species for the production of ITK formulations to control pests (Sharma 2021). These herbs have prominence in tea plantations as weeds have traditionally been used in folk medicine and have shown antimicrobial and anti-inflammatory properties. Some of the plants that are popularly used as botanicals are Clerodendrum viscosum Vent.,Melia azadirachta L., Artimisia vulgaris L.,Cassia tora L., Justicia adhatoda (L.) Nees, Polygonum hydropiper L., etc. Among these, Justicia adhatoda (L.) Nees (family Acanthaceae) shows a wide range of activities including antimicrobial, anti-inflammatory, anticholinesterase, abortifacient, cardioprotective, hepatoprotective, and antitussive properties. The major alkaloids of the plant, Vasicine and Vasicinone, are biologically active and are used in pharmacological and toxicological studies (Dhankhar et al. 2011). When compared to controls, J. adhatoda extracts lowered fertility and oviposition in gravid females of Aedes aegypti L. (Thanigaivel et al., 2017). This is in concordance with the previous report where Justicia adhatoda has been documented to be an effective managerial agent against pests (Williges 2004). Similarly, Polygonum hydropiper (L.) (family Polygonaceae) leaf oils exhibit neuroprotective potentials primarily due to their antiradical and cholinesterase activities (Tong et al., 2021). The hot water extract of Polygonum hydropiper is an effective anti-feedant against bean aphids Aphis craccivora with mortality ranging from 87.6- 94.5% with P < 0.01 after a week since spray application (Das et al. 2008). A compound “Polygodial” a compound is an active antifeedant against Egyptian or African cotton leafworms (Spodoptera littoralis), whiteflies (Bemisia tabaci), and various aphids (such as Aphis craccivora, Myzus persicae, Rhopalosiphum padi) (Asakawa et al. 1988; Prota et al. 2013). There is not much work regarding the efficacy of Justicia adhatoda (L.) Nees and Polygonum hydropiper L. against Oligonychus coffeae Neitner have been found to be done in India. Therefore, the present work has been designed to primarily evaluate the efficacy of aqueous extracts of J. adhatoda and P.hydropiper to control the tea red spider mite (O.coffeae) as a part of integrated pest management with inquisitive emphasis on adulticidal and ovicidal effect.
Materials and methods
Red Spider Mite (RSM) rearing
Red Spider Mite-infested bushes from the bio-organically maintained tea garden of the University of North Bengal, India were identified and mite-infested leaves were collected. A pair of closely spaced moist cotton rectangular strips was placed along the length of the laboratory polypropylene utility tray. Fresh leaves from the same mite-infested plants were collected. The fresh leaves were cleaned carefully with distilled water and dried for a short time, and then both the infested and fresh leaves’ petioles were wrapped with pieces of moist cotton and all the leaves were placed on the cotton padding in the tray with their upper surfaces facing upward as mites favour the upper surface of leaves and not the lower. The whole setup was sprayed with water subsequently to maintain the humidity of the leaves and keep them fresh (Figure 1). Sometimes mites were also transferred to fresh garden-collected tea from infested tea leaves using a fine camel hair brush so to facilitate their dispersion and increase their population. Fresh leaves from susceptible clones were introduced for four days.
Figure 1: Tea infected with Oligonychus coffeae (RSM), (a) Plant, (b) Single leaf, and (c) Rearing of RSM
Microscopy of RSM
A bright field binocular compound microscope (Labomed LX300) was used to identify mites at various magnifications as needed. Scanning electron microscopy (SEM) was carried out at the University Science Instrumentation Center (USIC) of the University of North Bengal, India. The images are depicted in figure 2.

Figure 2: (a) Oligonychus coffeae-Female (10X), (b) Oligonychus coffeae–Male (10X), and (c) SEM image of Oligonychus coffeae (female)
Preparation of botanicals
Aqueous extracts of each of the plants were prepared using the method described in the handbook (Anjarwalla and Belmain, 2016; Sharma 2021) with slight modifications according to the availability of resources. The collected plants were dried under shade for 30 days in the laboratory. Following this, each of the leaves of Justicia adhatoda and the leaves and stems of Polygonum hydropiper was ground using a grinder. The coarse powder thus obtained was sieved with a mesh to maintain uniformity of the particle size which would ensure that a uniform surface area of the plant powder particles is provided for the maximum extraction. 100 g of both the plant powders were weighed and poured into 1 L capacity conical flasks each with 4 g of finely chopped soap nut (Sapindus mukorossi). The stock solutions of both plants were prepared in this manner. These solutions were macerated for 48 hours in dark with intermittent stirring for extraction of the active principles from the fine powders of the aerial parts of the plants used. The crude extracts were filtered using clean pieces of muslin clothes and used as stock solutions to prepare the rest of the different concentration strength aqueous extracts of Polygonum hydropiper, and Justicia adhatoda.
Bioassay studies of botanical aqueous extracts on Oligonychus coffeae adults
For the current bioassay on determining the mortality and median lethal dose of aqueous extract on tea red spider mite, the leaf-disc technique was employed (Banerjee et al., 2021; Mitra et al. 2015). All the assays were conducted under laboratory conditions. The leaf discs were cut out of the tea clones TV25 and TV26, both of which are found to be susceptible to RSM. Tea leaf discs of 2 cm diameter were dipped in different concentrations of each of Polygonum hydropiper and Justicia adhatoda for 5 minutes and then dried. These leaf discs were laid with their adaxial surface up on moist cotton beds or moist filter paper in mini petri dishes. Twenty moving adult mites were transferred on each treated tea leaf disc using a fine camel hair brush. Water-treated leaf discs were used as a control. The assay was replicated three times for each concentration of the studied plants. The leaf discs were observed after 24 and 48 hours (Figure 3). Immobility in mites when prodded with fine camel hair brush after 24 hours and 48 hours from transfer suggested mortality. Furthermore, the mortality was confirmed by inspecting the mites under a bright field binocular compound microscope (Labomed LX300).

Figure 3: (a) The leaf disc cut out being dipped into aqueous extract and (b) The treated leaf disc transferred on moist filter paper
Statistical analysis
LC50 values, or lethal concentrations that resulted in 50% larval death, were discovered using Probit analysis and IBM SPSS version 21. Results with a p ≤ 0.05 threshold were regarded as statistically noteworthy (Poddar et al. 2013; Subba et al.2020).
Ovipositional deterrence
The ovipositional deterrence caused by aqueous extract after 48 hours were studied by following the method of (Roobakkumar et al. 2010; Roy et al. 2011), and the discrimination quotient was calculated using the formula given in (Roobakkumar et al. 2010) using MS Excel. Five gravid females were transferred on each of the variously treated leaf discs and were allowed to oviposit. The number of eggs laid was counted after 48 hours of transfer of all the mites. All the experiments have been done by taking water-treated leaf discs as a control. The formula used for calculating the discrimination quotient is according to (Roobakkumar et al. 2010):
Where, T = treated mites and C = mites in control.
Results
Bioassay studies of botanical aqueous extracts
Adult O. coffeae mortality was linearly related to the time elapsed after treatment for each increasing concentration by probit analysis. After 48 hours have elapsed the LC50 values for J. adhatoda aqueous extract is 36.895 mg/mL, P. hydropiper aqueous extract is 50.04 mg/mL, and LC90 values for J. adhatoda is 95.525 mg/ml. In the case of control, mortality was null concerning J. adhatoda and P. hydropiper (Figures 4 and 5). According to the experiment, aqueous extract of J. adhatoda shows that the least LC50 and LC90 values are among the two (Figure 6). Thus, the aqueous extract of J. adhatoda is comparatively the most toxic among the two used aqueous extracts.

Figure 4: (a) Polygonum hydropiper probit analysis and (b) Justicia adhatoda probit analysis

Figure 5: Mortality rate of O. coffeae with the treatment of botanicals
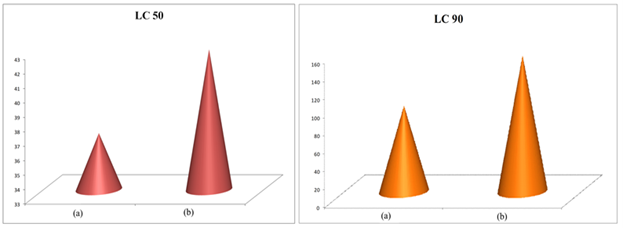
Figure 6: LC50 and LC90 value for (a) Justicia adhatoda and (b) Polygonum hydropiper
Ovipositional deterrence
The botanicals showed ovipositional deterrence and are measured by discrimination quotient (DQ). The discrimination quotient ranges from 0 to 1.0 and is a tool for determining the effect of chemicals on the egg-laying behaviour of insects. When the leaves were treated with the botanicals, the mites discriminated among the treated leaves concerning the number of eggs laid. Red spider mites did not lay eggs on leaf discs treated with different concentrations of aqueous extract of the botanicals. Ovipositional deterrence and discrimination quotient value is listed in Table 1.
Table 1: Ovipositional deterrence and Discrimination Quotient value of different botanicals on Oligonychus coffeae
|
Sl. No |
Treatment |
Dosage mg/mL |
No. of eggs laid (mean) |
DQ value |
|
1 |
Justicia adhatoda |
25 |
21.3 |
0.169 |
|
50 |
16.7 |
0.284 |
||
|
75 |
8.2 |
0.57 |
||
|
100 |
1.0 |
0.935 |
||
|
2 |
Polygonum hydropiper |
25 |
7.4 |
0.604 |
|
50 |
6.2 |
0.657 |
||
|
75 |
4.0 |
0.764 |
||
|
100 |
2.0 |
0.9 |
||
|
3 |
Control (water) |
_ |
30 |
_ |
Discussion
The mite populations of Terai and Dooars showed significant tolerance to conventionally used acaricides such as Dicofol and Ethion because they being cheap were in use for a significant period of time. However, resistance to pesticides in arthropod pests was observed may be due to increased detoxification or metabolism of the pesticide toxins, decreased rate of cuticular penetration, and elevated sequestration levels of the toxins (Roy, 2019). This has turned attention to the use of botanicals as pesticides. Thus with increasing global awareness, the application of conventional chemical pesticides is seeing its fall daily. The present bioassay and ovipositional deterrence studies were done using pieces of tea leaves dipped in different concentrations of aqueous extracts of J. adhatoda and P.hydropiper on O. coffeae (RSM). The aqueous extract of J.adhatoda shows the least LC50 and LC90 values and the P. hydropiper aqueous extract shows the highest LC50 value J. adhatoda is reported to lower fitness, damage the mid-gut cells, and block enzymes that protect from oxidative damage in Aedes aegypti (Thanigaivel et al. 2017). While studying ovipositional deterrence, the number of mite eggs laid on leaf discs treated with the highest aqueous extract of J. adhatoda was zero and the DQ value it furnished was 1.0, and thus is consistent with the findings of (Thanigaivel et al. 2017) on the anti-oviposition effect of J. adhatoda on the gravid females of the Aedes aegypti , P.hydropiper has also exhibited the similar results.
As for the cost effectiveness is concerned, the cost of a technical grade acaricide is somewhere around INR 450.00 in India, but the cost of preparing botanicals from commonly growing weeds in the tea plantation is almost nil other than the labour charges. Therefore, economically, botanicals can also reduce the cost load of controlling red spider mites at least impeding them from reaching the economic threshold level. Botanicals would be expected to have an upper hand in preventing pesticide resistance in mites also because a wide range of active compounds are sprayed and not a single constituent is derived through a series of industrially operated extraction techniques. However, the problem with botanicals is that they should be sprayed during dusk as the botanicals have photolabile compounds that will degrade under the sun therefore any botanical application is recommended to be sprayed during dusk for the active principles to work throughout the period when it is dark up until dawn (Anjarwalla et al. 2016).
Conclusion
The aqueous extracts chosen were found to possess significant acaricidal and ovipositional deterrent properties. Even though both extracts are effective on the adults of O.coffeae, the aqueous extract of J.adhatoda showed a better pesticidal effect. This study validates the acaricidal property and ovipositional deterrent properties of the aqueous extract of the plants. However, they were carried out under laboratory conditions, and therefore, their field efficacy remains to be seen in the plantations of the North Bengal area under the current climatic changes.
Acknowledgments
AS acknowledges Biswa Bangla Genome Center, University of North Bengal.
Conflict of interest
The authors declare they have no competing interests.
Copyright © 2023 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
|
Citation Arnab Sen*, Sandipan Ghosh, Soumyadip Sengupta, Gargi Sen, Chandra Ghosh, Acaricidal and Ovicidal effects of two common weeds of tea gardens of North Bengal, India against Oligonychus coffeae.Journal of research im weed science, 2023, 6(2), 40-49. http://dx.doi.org/10.26655/JRWEEDSCI.2023.6.5
|
| Article View | 897 |
| PDF Download | 376 |